 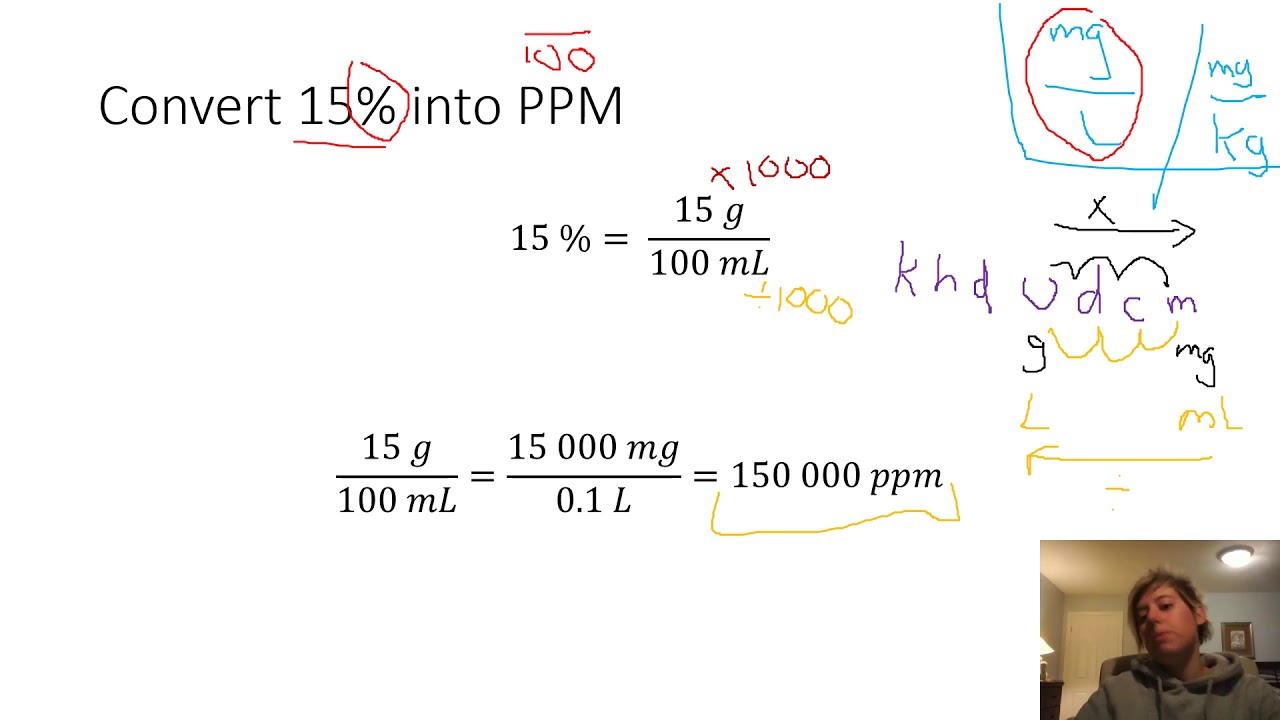
Conversion from mass concentration to ppmīy dividing the mass of gas in a cubic meter by the molar mass of gas, we can get the number of moles of gas in a cubic meter. Finally, by multiplying the number of moles of gas by the molar mass of gas, we can obtain the mass of gas in one cubic meter of total volume or mass concentration. By dividing this number by the molar volume of the gas (for a given temperature and pressure), we can get the number of moles of gas in one cubic meter of total volume. 
Knowing ppm, we can find how many liters of gas are contained in one cubic meter of total volume. 
Where the universal gas constant R ≈ 8.314 J / (mol K) Conversion from ppm to mass concentration You can find the molar volume for a given temperature and pressure using the Clapeyron equation: According to the first corollary from Avogadro's law, one mole of any gas under the same - isobaric and isothermal - conditions occupies the same volume. The conversion from one unit to another is based on the Law of Avogadro's, according to which equal volumes of gases, at the same temperature and pressure, contain the same number of molecules. For example, permissible exposure limits (PEL or OSHA PEL) are expressed both in ppm and in mg/m³. Sometimes, however, the gas concentration is expressed as the mass concentration in milligrams per cubic meter (mg/m3). By definition, it is the number of gas particles per million air particles, 1 ppm = 1/1000000 = 0.0001% = 0.001‰. Ppm is often used for low concentration values, typical for air pollution. or ppmv (parts per million by volume), although IUPAC recommends avoiding such notation 1. Sometimes, to emphasize that we are talking about volume concentration, you can see %vol. It is a dimensionless value, for convenience expressed in percent (%), permille (parts per thousand, ‰), or ppm (parts per million). Conversion from mass concentration to volume concentration and vice versaĪs mentioned above, the volume concentration of gas is defined as the ratio of the gas volume to the total volume. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
